4DMT seeks to unlock the full potential of genetic medicines using its platform, Therapeutic Vector Evolution, which combines the power of directed evolution with approximately one billion synthetic capsid sequences to invent targeted and evolved vectors for use in our products. This increase in net loss was primarily driven by increased research and development expenses associated with the progression of our clinical product candidates, including 4D-150 for wet AMD, 4D-310 for Fabry disease and 4D-710 for cystic fibrosis lung disease.ĤDMT is a clinical stage biotherapeutics company harnessing the power of directed evolution for targeted genetic medicines. 
Net Loss: Net loss was $25.7 million for the quarter ended September 30, 2022, as compared to $22.2 million for the quarter ended September 30, 2021. G&A Expenses: General and administrative expenses were $8.1 million for the quarter ended September 30, 2022, as compared to $8.2 million for the quarter ended September 30, 2021. This increase was primarily driven by the progression of our clinical product candidates, including 4D-150 for wet AMD, 4D-310 for Fabry disease and 4D-710 for cystic fibrosis lung disease. R&D Expenses: Research and development expenses were $18.9 million for the quarter ended September 30, 2022, as compared to $15.8 million for the quarter ended September 30, 2021. Revenue: Total revenue for the quarter ended September 30, 2022, was $0.5 million, as compared to $1.4 million for the quarter ended September 30, 2021. Kirn added, “During 2022, we have maintained our discipline and focus on efficient cash utilization with current cash available to support our planned operations into the first half of 2025.” We look forward to further patient assessments, and to enrolling additional patients onto the 4D-710 clinical trial to continue the progress of our 4D-710 product candidate,” said David Kirn, M.D., Co-founder, and Chief Executive Officer of 4DMT. 
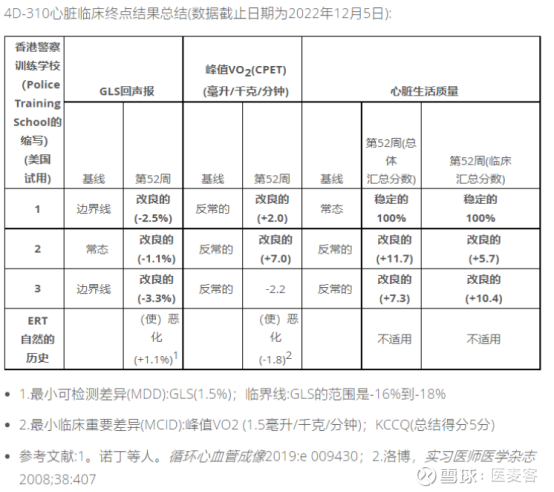
These clinical data illustrate the potential of this platform to invent optimized vectors for our genetic medicines in multiple therapeutic areas. We have now demonstrated potential safety, tolerability, and clinical activity with three different proprietary synthetic AAV-derived vectors, each delivered by a routine route of administration, that we invented through our Therapeutic Vector Evolution platform. "We are encouraged by the interim clinical data we have released from our aerosol delivered 4D-710 program for the treatment of cystic fibrosis lung disease. The 4D-710 product candidate comprises the proprietary synthetic aerosol delivered vector A101 that was invented at 4DMT. Levels of transgene expression were in a range that was predicted to be associated with clinical benefit. Analyses of 11 total lung biopsies and brushings (Week 4) demonstrated widespread delivery and expression of the 4D-710 CFTRΔR transgene in all samples across all three patients’ lungs. 
On November 3, 2022, interim clinical data from patients enrolled in cohort 1 of 4DMT’s Phase 1/2 clinical trial of 4D-710 for the treatment of cystic fibrosis lung disease was presented at the North American Cystic Fibrosis Conference, which demonstrated safety and tolerability with no 4D-710-related adverse events following aerosol delivery. (Nasdaq: FDMT), a clinical-stage biotherapeutics company harnessing the power of directed evolution for targeted genetic medicines, today reported third quarter 2022 financial results. 09, 2022 (GLOBE NEWSWIRE) - 4D Molecular Therapeutics, Inc. Cash, cash equivalents and marketable securities sufficient to fund operations into the first half of 2025ĮMERYVILLE, Calif., Nov. Interim clinical trial data from 4DMT’s Phase 1/2 clinical trial of 4D-710 for the treatment of cystic fibrosis lung disease was presented at North American Cystic Fibrosis Conference on November 3, 2022 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
